
S.M.A.R.T. CONTROL Vascular Stent System
When outcomes matter, design matters. For superficial femoral artery (SFA) and iliac lesions.
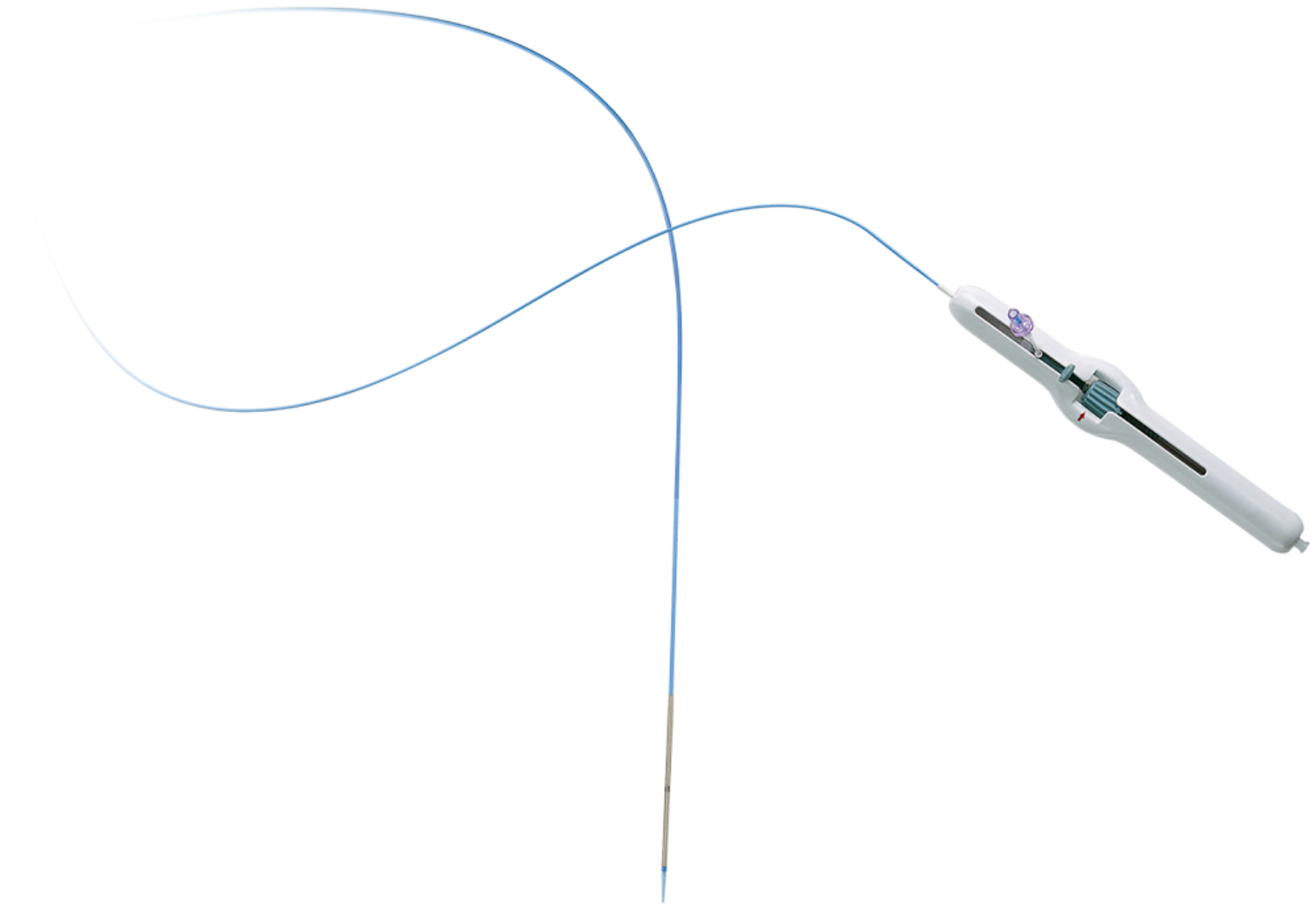
Product Description
The S.M.A.R.T. CONTROL® Vascular Stent System offers:
- Longitudinal stability: Greater stability minimizes stretching at deployment, thereby increasing placement accuracy.
- Uniform scaffolding: Smaller cell size and uniform coverage can help prevent vessel prolapse.
- Radial force: The stent’s ability to resist compression maintains luminal gain.
- Low fracture rate: The stent maintained a low fracture rate out to 3 years in the STROLL Study with the S.M.A.R.T. Vascular Stent Systems.1
1. Cordis Data on file.
Product Characteristics
Scroll, sort or search for the particular S.M.A.R.T. CONTROL Vascular Stent System's part number of interest.
| SKU | Product Description | Sheath Compatibility (F) | Guidewire compatibility (in) | Stent Diameter (mm) | Stent Length (mm) | Shaft Length (cm) |
|---|---|---|---|---|---|---|
| C10060SL | SMART CONTROL STENT 10 X 60 X 80CM | 6 | 0.035 | 10 | 60 | 80 |
| C10060ML | SMART CONTROL STENT 10 X 60 X 120CM | 6 | 0.035 | 10 | 60 | 120 |
| C10040SL | SMART CONTROL STENT 10 X 40 X 80CM | 6 | 0.035 | 10 | 40 | 80 |
| C10040ML | SMART CONTROL STENT 10 X 40 X 120CM | 6 | 0.035 | 10 | 40 | 120 |
| C10030SL | SMART CONTROL STENT 10 X 30 X 80CM | 6 | 0.035 | 10 | 30 | 80 |
| C09060SL | SMART CONTROL STENT 9 X 60 X 80CM | 6 | 0.035 | 9 | 60 | 80 |
| C09060ML | SMART CONTROL STENT 9 X 60 X 120CM | 6 | 0.035 | 9 | 60 | 120 |
| C09040SL | SMART CONTROL STENT 9 X 40 X 80CM | 6 | 0.035 | 9 | 40 | 80 |
| C09040ML | SMART CONTROL STENT 9 X 40 X 120CM | 6 | 0.035 | 9 | 40 | 120 |
| C08100SL | SMART CONTROL STENT 8 X 100 X 80CM | 6 | 0.035 | 8 | 100 | 80 |
| C08100ML | SMART CONTROL STENT 8 X 100 X 120CM | 6 | 0.035 | 8 | 100 | 120 |
| C08080SL | SMART CONTROL STENT 8 X 80 X 80CM | 6 | 0.035 | 8 | 80 | 80 |
| C08080ML | SMART CONTROL STENT 8 X 80 X 120CM | 6 | 0.035 | 8 | 80 | 120 |
| C08060SL | SMART CONTROL STENT 8 X 60 X 80CM | 6 | 0.035 | 8 | 60 | 80 |
| C08060ML | SMART CONTROL STENT 8 X 60 X 120CM | 6 | 0.035 | 8 | 60 | 120 |
| C08040SL | SMART CONTROL STENT 8 X 40 X 80CM | 6 | 0.035 | 8 | 40 | 80 |
| C08040ML | SMART CONTROL STENT 8 X 40 X 120CM | 6 | 0.035 | 8 | 40 | 120 |
| C08030SL | SMART CONTROL STENT 8 X 30 X 80CM | 6 | 0.035 | 8 | 30 | 80 |
| C07100SL | SMART CONTROL STENT 7 X 100 X 80CM | 6 | 0.035 | 7 | 100 | 80 |
| C07100ML | SMART CONTROL STENT 7 X 100 X 120CM | 6 | 0.035 | 7 | 100 | 120 |
| C07080SL | SMART CONTROL STENT 7 X 80 X 80CM | 6 | 0.035 | 7 | 80 | 80 |
| C07080ML | SMART CONTROL STENT 7 X 80 X 120CM | 6 | 0.035 | 7 | 80 | 120 |
| C07060SL | SMART CONTROL STENT 7 X 60 X 80CM | 6 | 0.035 | 7 | 60 | 80 |
| C07060ML | SMART CONTROL STENT 7 X 60 X 120CM | 6 | 0.035 | 7 | 60 | 120 |
| C07040SL | SMART CONTROL STENT 7 X 40 X 80CM | 6 | 0.035 | 7 | 40 | 80 |
| C07040ML | SMART CONTROL STENT 7 X 40 X 120CM | 6 | 0.035 | 7 | 40 | 120 |
| C07030ML | SMART CONTROL STENT 7 X 30 X 120CM | 6 | 0.035 | 7 | 30 | 120 |
| C07020ML | SMART CONTROL STENT 7 X 20 X 120CM | 6 | 0.035 | 7 | 20 | 120 |
| C06100SL | SMART CONTROL STENT 6 X 100 X 80CM | 6 | 0.035 | 6 | 100 | 80 |
| C06100ML | SMART CONTROL STENT 6X100 X 120CM | 6 | 0.035 | 6 | 100 | 120 |
| C06080SL | SMART CONTROL STENT 6 X 80 X 80CM | 6 | 0.035 | 6 | 80 | 80 |
| C06080ML | SMART CONTROL STENT 6 X 80 X 120CM | 6 | 0.035 | 6 | 80 | 120 |
| C06060SL | SMART CONTROL STENT 6 X 60 X 80CM | 6 | 0.035 | 6 | 60 | 80 |
| C06060ML | SMART CONTROL STENT 6 X 60 X 120CM | 6 | 0.035 | 6 | 60 | 120 |
| C06040SL | SMART CONTROL STENT 6 X 40 X 80CM | 6 | 0.035 | 6 | 40 | 80 |
| C06040ML | SMART CONTROL STENT 6 X 40 X 120CM | 6 | 0.035 | 6 | 40 | 120 |
| C06030SL | SMART CONTROL STENT 6 X 30 X 80CM | 6 | 0.035 | 6 | 30 | 80 |
| C06030ML | SMART CONTROL STENT 6 X 30 X 120CM | 6 | 0.035 | 6 | 30 | 120 |
| C06020ML | SMART CONTROL STENT 6 X 20 X 120CM | 6 | 0.035 | 6 | 20 | 120 |

PRODUCTS YOU MAY ALSO BE INTERESTED IN
IFU
Please refer to the Instructions for Use for complete information, including indications, precautions, warnings, and potential adverse events.
Customer Service and Ordering Information
In the United States, email us your question or order, or call us at 800.327.7714.



