
OUTBACK Elite Re-entry Catheter
The OUTBACK® Elite Re-entry Catheter enables the re-entry of a guidewire from the subintimal space back into true lumen of the vessel.
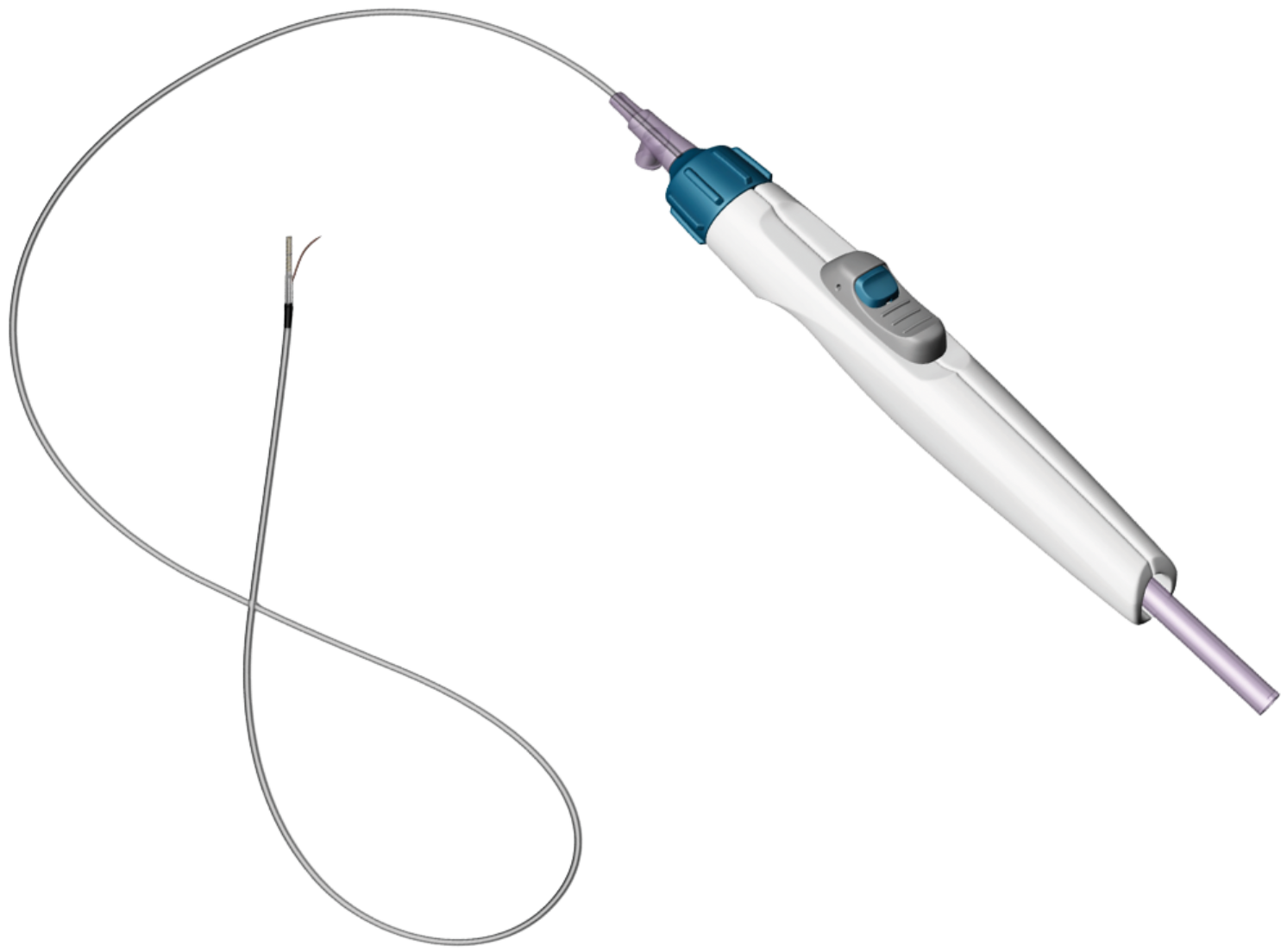
Product Characteristics
Scroll, sort or search for the particular OUTBACK ELITE Re-entry Catheter's part number of interest.
| SKU | Product Description | Sheath Compatibility (F) | Catheter Length (cm) | Shape | Guidewire Compatibility (in) |
|---|---|---|---|---|---|
| OTB59120A | OUTBACK ELITE RE-ENTRY CATHETER 6F 120CM | 6 | 120 | NA | n/a |
| OTB59080A | OUTBACK ELITE RE-ENTRY CATHETER 6F 80CM | 6 | 80 | NA | n/a |
Resources
IFU
Please refer to the Instructions for Use for complete information, including indications, precautions, warnings, and potential adverse events.
Customer Service and Ordering Information
In the United States, email us your question or order, or call us at 800.327.7714.

